Taking a humane look at cosmetics
Updated: 2013-12-20 08:48
By Xu Wei and Zhang Lei in Beijing (China Daily)
|
|||||||||||
|
|
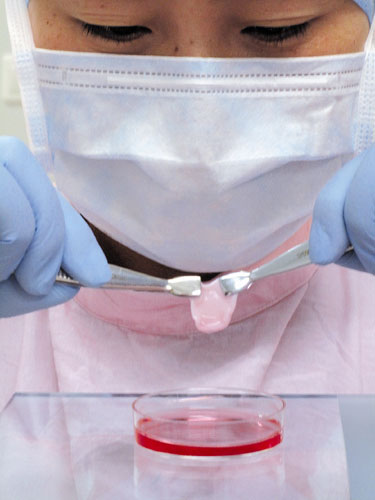
Strict safety tests
The Hygienic Standards for Cosmetics, China's guidelines for safety tests on cosmetics, were introduced by the former Ministry of Health in 2007.
The methods specified by the regulation, which is based on guidelines drawn up by the Organization for Economic Cooperation and Development, require that all cosmetic products be subjected to 17 animal-based toxicological tests, such as those for acute oral toxicity, acute eye irritation, skin sensitization and a combined test for chronic toxicity and carcinogenicity.
The skin sensitization test uses 20 guinea pigs as samples and a further 10 as "controls", that is, they don't undergo the tests. Cosmetics are repeatedly applied to a shaved area on the subject animal's back, the condition of the skin is then compared with its control and any changes are noted.
In the combined chronic toxicity/carcinogenicity test, both the test and control groups comprise 100 animals, a 50-50 split of males and females. The tests are conducted continually throughout the animals' life spans, usually about a year.
Li Hua, president of Animal Guardians, a nongovernmental animal rights group, said the problems associated with the protection of lab animals stem from the fact that the general public knows little about what happens in the labs.
"Even animal right activists such as my organization are unable to gain access to inside information, so the industry is effectively closed to outsiders," she said.
Improvements in animal welfare were first introduced by the Ministry of Science and Technology, which issued China's first guidelines on the humane treatment of lab animals -including advice on breeding, transportation and the conduct of the experiments - in October 2006.
Since then, the authorities in a number of areas, including the provinces of Guangdong and Hubei, and Beijing, have formulated their own regulations.
Compared with the laws and regulations in Western countries, though, some species have been omitted from the list covered by the test regulations, according to He Zhengming, a researcher with the National Institutes for Food and Drug Control.
In an article published in 2011, he noted that the national and local standards include the most-commonly used species, but fail to cover animals such as Mongolian gerbils and domestic cats.
Today's Top News
Nearly 90 hurt as London theatre ceiling collapses
Putin to pardon jailed tycoon
More help on way for foreign media
Beijing vows to boost Bolivia infrastructure
'Mutual interests beat differences'
New envoy 'must find right mix'
Yanukovich offers protesters nothing
China, Britain face new opportunities
Hot Topics
Lunar probe , China growth forecasts, Emission rules get tougher, China seen through 'colored lens', International board,
Editor's Picks

|

|

|

|

|

|